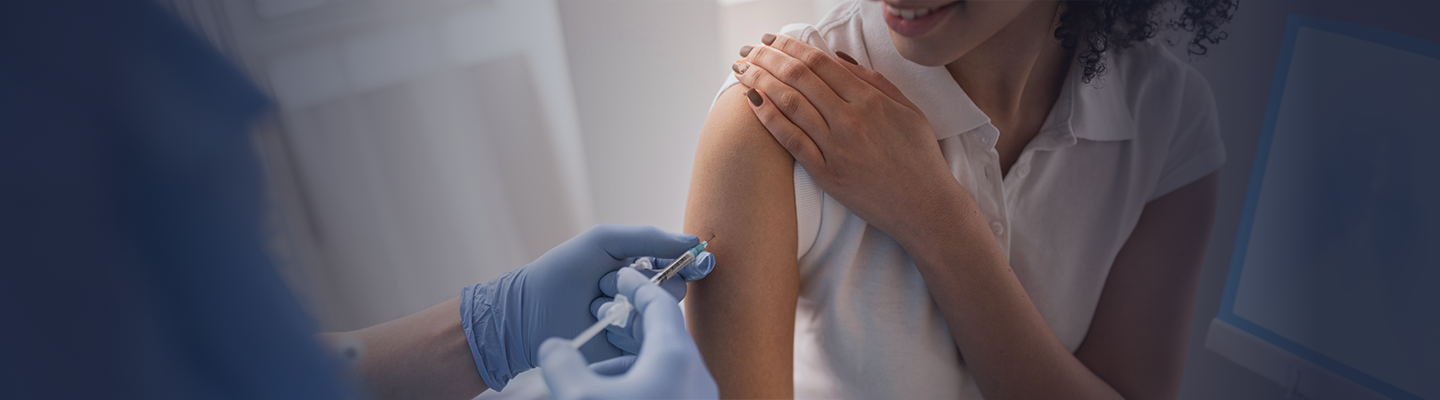
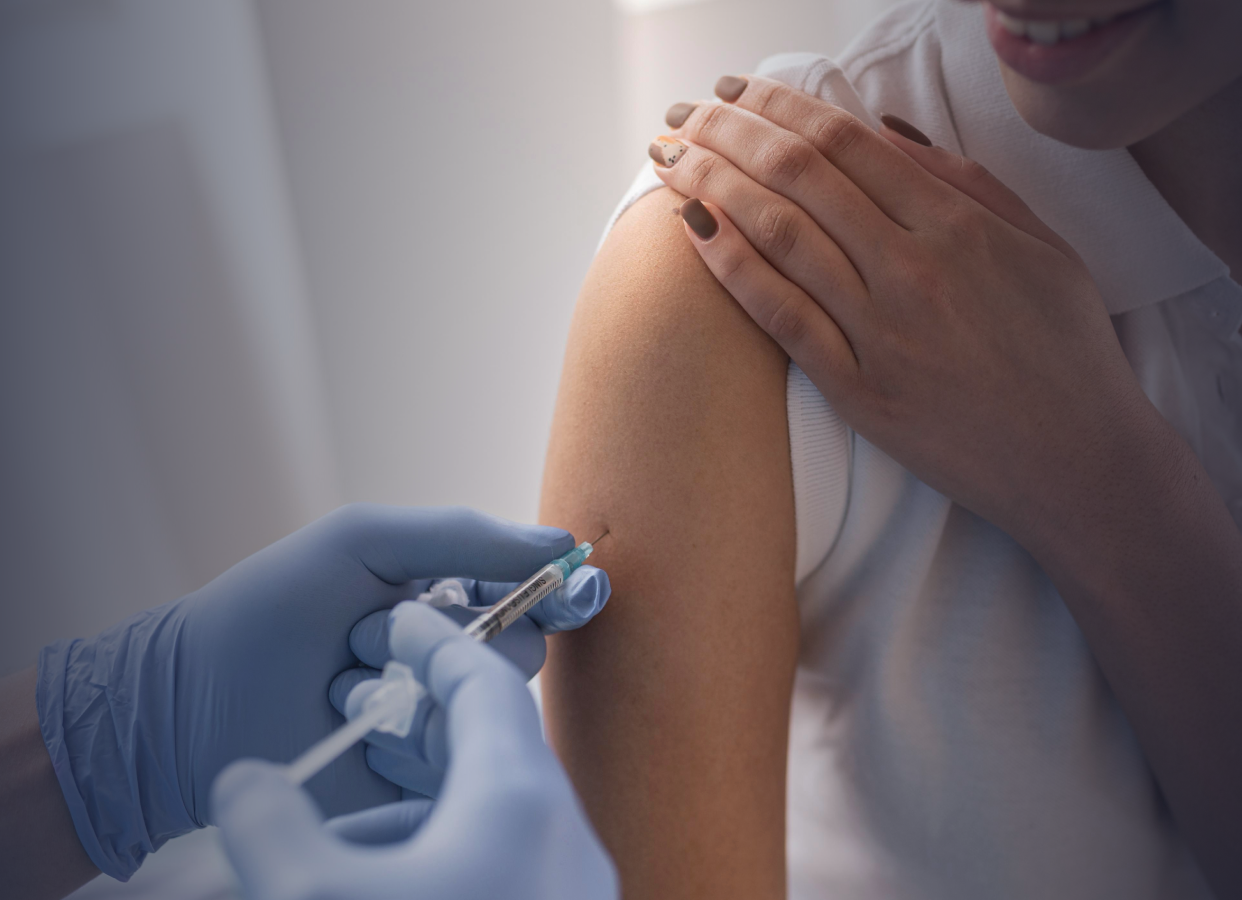
What We Do
Hilleman Laboratories operates a first-of-its-kind vaccine development hub in Singapore, consisting of a research and development (R&D) facility at Biopolis, alongside a 30,000 square foot pilot-scale manufacturing facility at 138 Depot Road. The hub provides end-to-end product development solutions from concept to pilot manufacturing and Phase II clinical studies, supporting the development of affordable, high-value vaccines and biologics for low- and middle-income countries.
Research and Development
The state-of-the-art R&D facility in Biopolis undertakes process development, analytical development, drug product development and formulation for vaccines and biologics. It focuses on product development of vaccines and biologics, including candidate selection, design, early manufacturing process development and preclinical studies.

GMP Pilot Manufacturing
As one of the facilities of the dual-feature set up, the Good Manufacturing Practices (GMP) facility is capable of producing clinical trial material (up to Phase II) and is equipped with drug product fill-&-finish capability. This unique set-up enables the two facilities working in tandem to provide end-to-end product development, leveraging innovative technology platforms to develop affordable, high-value vaccines and biologics. The facility has also been designed to easily pivot to manufacture vaccines for emergency use during pandemics.

Partnerships
Hilleman is a critical connector, bringing together health ecosystem stakeholders for vaccines and biologics R&D and manufacturing. We collaborate with local, regional and global partners and stakeholders, including policymakers and governments, to facilitate wider, affordable access to life-saving vaccines and biologics. Apart from conducting translational research and product development, we also act as a catalyst for advancements in the biopharma industry by facilitating technology transfers, and capability building through manpower development initiatives.
Our Partners